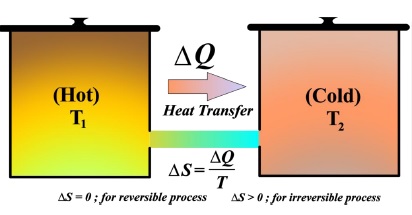
This course provides a foundation in thermodynamics, beginning with the concept of temperature and the Zeroth Law, which establishes thermal equilibrium and underpins temperature and pressure measurements. Students then explore how work and heat contribute to energy changes in a system, leading into the First Law of Thermodynamics (conservation of energy) and its applications to ideal gases. The Second Law introduces the concept of irreversibility and states that the total entropy of an isolated system never decreases and sets limits on the efficiency of heat engines—illustrated by the Carnot cycle. Applications for heat engine (as steam, gasoline, and diesel heat engines), refrigerator and heat pump through evaluation of their efficiency and coefficient of performance, respectively. Overall, these concepts form the foundation of thermodynamics, illustrating how energy is exchanged and conserved in physical processes, how equilibrium is defined and measured, and how entropy governs the direction of spontaneous processes. The course also covers thermodynamic potentials, Maxwell’s relations, which help describe processes under various conditions.
- Formator: AURELIA-ANCA DUMITRU